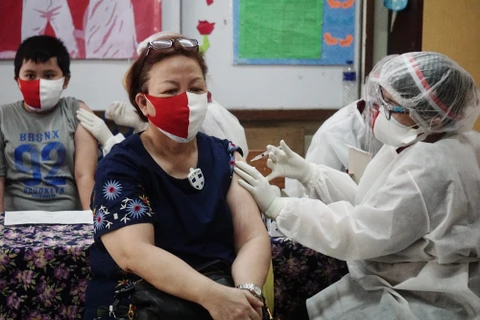
Hanoi (VNA) – The Food and Drug Administration (FDA) of the Philippines has approved the use of Moderna’s COVID-19 vaccine for children aged six to 11 years old, according to the country’s Department of Health (DOH).
The DOH said on May 31 that the decision was issued on May 20, but evaluation and a positive recommendation from the Health Technology Assessment Council will be needed before the use of the vaccine is rolled out.
On May 30, Zuellig Pharma Corporation said two doses of Moderna vaccine have “an acceptable safety profile” and can create strong immune response for children aged 6 to 11.
The corporation also pointed out that the efficacy of Spikevax among the younger age group is similar to those in adults.
In late December of 2021, the Philippine FDA approved the emergency use authorization (EUA) for the Pfizer COVID-19 vaccine for children ages 5 to 11 years old./.
The DOH said on May 31 that the decision was issued on May 20, but evaluation and a positive recommendation from the Health Technology Assessment Council will be needed before the use of the vaccine is rolled out.
On May 30, Zuellig Pharma Corporation said two doses of Moderna vaccine have “an acceptable safety profile” and can create strong immune response for children aged 6 to 11.
The corporation also pointed out that the efficacy of Spikevax among the younger age group is similar to those in adults.
In late December of 2021, the Philippine FDA approved the emergency use authorization (EUA) for the Pfizer COVID-19 vaccine for children ages 5 to 11 years old./.
VNA