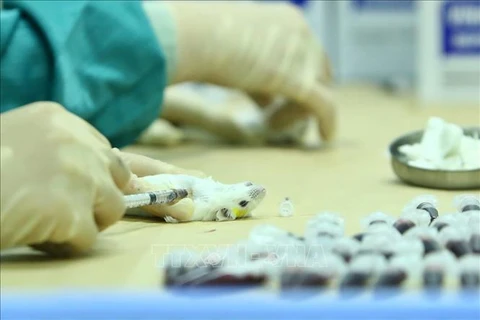
Hanoi (VNS/VNA) - Vietnam has started human trials of a COVID-19 vaccine.
Volunteers were recruited for the first phase of the trials of a vaccine on December 10 in Hanoi.
The vaccine was produced by the Nanogen Pharmaceutical Biotechnology company, one of four organisations researching COVID-19 vaccines in Vietnam.
Speaking at the recruitment ceremony, Nguyen Ngo Quang, deputy director of the Ministry of Health’s Administration of Science Technology and Training, said: “The core purpose of the first phase of human trials is to evaluate the safety of a vaccine when used on humans, not its effectiveness, potency, or ability to provoke an immune response in the body.
“So our main objective for this first phase is to identify the optimal dose so we can move on to the second phase.
“The second and third phases are when we evaluate the vaccine’s immunogenicity on a wide population.”
The ceremony was co-organised by the Ministry of Health and the Military Medical Academy.
The academy is also the venue where vaccine injections will take place.
“All accommodation, facilities and equipment are ready for the human trials at the academy. Everything has been prepared in accordance with standards required by the Ministry of Health,” said the academy’s director, Lieutenant General Do Quyet.
“We have prepared an isolated place for vaccine injections, volunteer monitoring and emergency aid.”
The volunteers will be injected with the first dose of the vaccine on December 17 and the second dose 28 days later, according to the health ministry.
Sixty volunteers are expected to be recruited for phase one, Quyet added.
“We will recruit 60 volunteers and divide them into three groups, each group will be testing a different dose of vaccine, from 25 to 50 to 75 micrograms,” he said.
“We put the safety of volunteers first. So in the preparation and selection processes, a health check is compulsory. And after they have been injected with the vaccine we will continue to monitor and be ready to handle unexpected situations.
“As the head of the clinical trial and research programme I can guarantee that if the vaccine is not safe we will stop the trials.
“If everything goes well, by the end of February next year we will have completed the first and second phases and move on to the third phase, which will have a lot more participants. If we succeed with the third phase, it is expected that by the end of 2021 the vaccine will be widely distributed.”
The volunteers registering for the trial on Thursday were mostly students at the academy. All of their personal information is kept confidential.
A 21-year-old male volunteer said: “The research of a COVID-19 vaccine has been going on for quite a long time in Vietnam. And the Ministry of Health has its own medical ethics committee, so I believe the vaccine is safe.
“I want to take part in the trials to prove its safety so more people will be injected and there won’t be a pandemic in the future.”
A 20-year-old female volunteer said: “I’m a student at the academy, so when I was informed about the human trials, I registered.
“I feel very nervous, because COVID-19 is a global pandemic, and vaccines are being widely tested around the world. Vietnam is one of the 42 countries which are developing the vaccines. I hope I can be one of the people who contribute to the creation of the vaccine.
“I really expect the vaccine will be effective. But I also think safety should always come first. I hope the vaccine will be safe, effective and reasonably priced.”/.
VNA