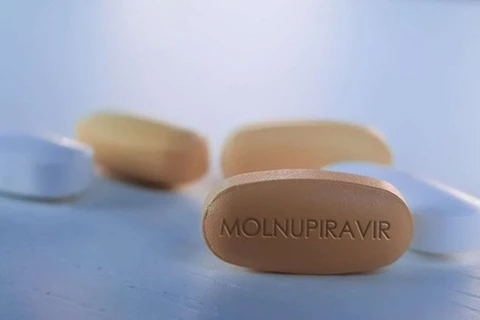
Packets of COVID-19 treatment drugs containing active ingredient Molnupiravir produced by Vietnamese manufacturers. (Photo: bostonpharma.com.vn)
Packets of COVID-19 treatment drugs containing active ingredient Molnupiravir produced by Vietnamese manufacturers. (Photo: bostonpharma.com.vn) Hanoi (VNA) – The Drug Administration of Vietnam (DAV) under the Ministry of Health (MoH) has announced the prices of COVID-19 treatment drugs containing active ingredient Molnupiravir produced by three Vietnamese manufacturers.
Accordingly, a pill of Molravir 400mg made by Boston Vietnam Pharma costs 11,550 VND while the prices of Movinavir 200mg manufactured by Mekophar Chemical Pharmaceutical, and Molnupiravir Stella 400mg produced by Stellapharm J.V Co., Ltd. are 8,675 VND and 12,500 VND a tablet, respectively.
These drugs are sold to COVID-19 patients for home treatment provided that they show proof of being infected with coronavirus and prescriptions from doctors.
The Drug Administration of Vietnam officially licensed the three COVID-19 drugs on February 17.
The DAV requested drug manufacturing and registration establishments to produce in accordance with dossiers and documents registered with the Ministry of Health, coordinate with treatment facilities to strictly comply with current regulations on prescription drugs, monitor the safety, effectiveness, and unwanted effects of drugs on Vietnamese people, and report it regularly.
The DAV also asked the Departments of Health of provinces and centrally-run cities to direct local medical examination and treatment facilities, medical staff, and drug supply establishments to notify patients of the benefits and risks when using these drugs, treatment methods, other drugs that can replace Molnupiravir in treating COVID-19.
During the circulation of the three drugs, based on the monitoring and updating information on the safety and effectiveness of them, the agency may decide to revoke the granted certificates of registration for circulation in line with Clause 1, Article 58 of the Law on Pharmacy.
These drugs are used to treat mild to moderate adult COVID-19 patients with at least one risk factor that could enhance the progression of the disease, according to the MoH.
The drugs should not be used for pregnant women or those who are planning to become pregnant, nursing women, and children under 18 years old./.