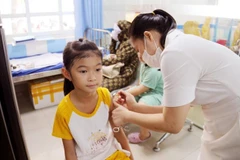
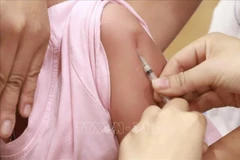
A medical worker gives a shot of Nano Covax to a volunteer in the third-phase trials at the Hanoi-based Vietnam Military Medical University (Photo: VNA)
A medical worker gives a shot of Nano Covax to a volunteer in the third-phase trials at the Hanoi-based Vietnam Military Medical University (Photo: VNA) Nguyen Ngo Quang, DeputyDirector of the Health Ministry’s Department for Science, Technology andTraining, made the remark in response to the NanogenPharmaceutical Biotechnology JSC’s proposal for validating its NanoCovax, a COVID-19 vaccine candidate, for emergency use.
Quang noted that beforeapproving a vaccine for mass use, it is a principle that the Ministry of Health(MoH) must ensure the safety, the generation of immunity, and the protectiveeffect of that vaccine.
The emergency useauthorisation for a vaccine depends on many factors, and the MoH needsscientific data to make a decision, Quang emphasised.
He stressed the MoH’sabsolute support for the development of domestic vaccines, adding that thehealth sector’s ultimate goal is to protect people’s health, so benefits andrisks must be thoroughly considered.
Nano Covax is currently inthe third phase of trials, with 1,000 volunteers already getting the shots.
Results recorded so farshowed that this vaccine candidate is safe and has generated the immuneresponse in volunteers, but its protective effect is still under research.
To have sufficient scientificbasis to assess the immunity generation and protective effect, volunteers needto be monitored for 36 days, 45 days, and 56 days since the first jabs,according to the official./.