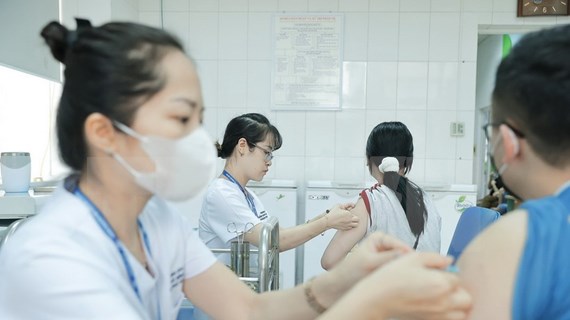
Vietnam’s two COVID-19 vaccines prove safe during trial
Two COVID-19 vaccines Nano Covax and Covivac, which were studied and developed in Vietnam, have proved to be safe and effective in the first and second clinical trials.
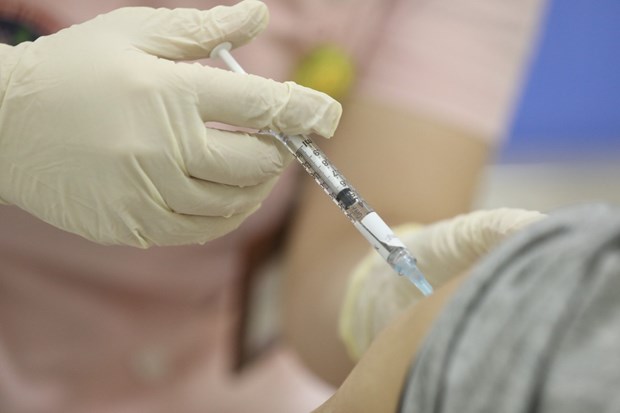 Illustrative image (Photo: VNA)
Illustrative image (Photo: VNA) Hanoi (VNA) – Two COVID-19 vaccines Nano Covax and
Covivac, which were studied and developed in Vietnam, have proved to be safe
and effective in the first and second clinical trials.
A total of 551 volunteers were given the second Nano Covax shots in the second stage, including 280 in Long An and 271 in Hanoi while 66 received the first Covivac shots in the first stage. The remaining nine in Hanoi will be vaccinated this week.
Nano Covax trial ahead of schedule
According to the Military Medical University, all volunteers receiving two Nano Covax shots in the second stage are in normal conditions. Only some of them suffered slight reactions such as swelling and muscle and joint pain, but the reactions quickly disappeared after resting.
At the end of the first clinical trial of Nano Covax, all participants were safe and produced a high concentration of antibodies. The vaccine proved effective on new variants of SARS-CoV-2 like those found in the UK.
In the current second stage, the vaccination is expanded to the elderly and those with underlying diseases. As many as 108 senior citizens were inoculated, with the oldest 76 years old. After vaccination, they had only slight reactions and showed no abnormal symptoms.
Results of the second stage of Nano Covax clinical trials are scheduled to be announced in late April, with optimistic prospects. The vaccine is expected to enter the third clinical trial in early May.
Based on results of two stages of trials so far, all experts said with optimism that the third stage will be completed at the end of the third quarter, three months ahead of the initial plan.
Covivac trial: first stage to end soon
Covivac, which was developed by the Health Ministry’s Institute of Vaccines and Medical Biologicals, is in the first stage of clinical testing by the Hanoi Medical University’s Centre of Clinical Pharmacology.
Since March 15, 66 volunteers have been vaccinated. Their slight reactions like swelling in injected area and headache disappeared within 24 hours after injection.
The remaining 54 volunteers will get the first shots from now to April 18.
The interval between the first and second shots is 28 days.
Six volunteers, who received the first shots on March 15, will become the first to be given the second shots on April 12./.